The world's leading system for automated high-level disinfection of ultrasound probes. Validated, guideline-compliant, fully documented — for patient safety without compromise.
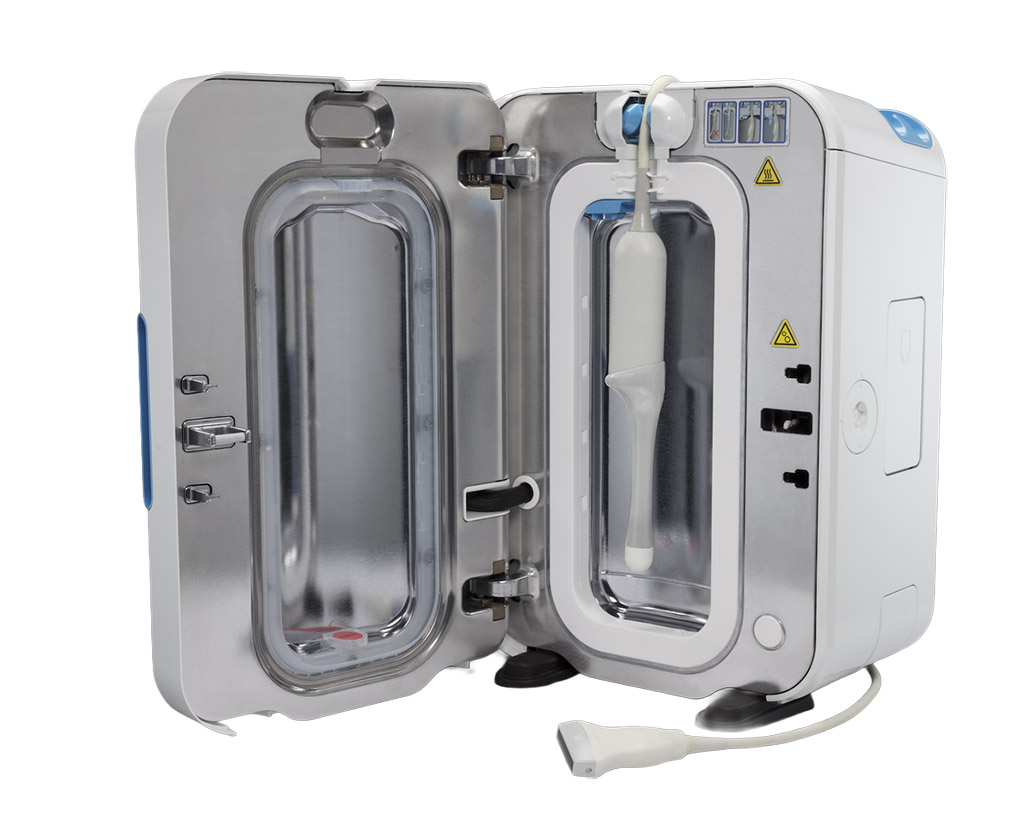
Ultrasound probes come into contact with mucous membranes on a daily basis — in gynaecology, anaesthesia, urology and cardiology. Manual disinfection is error-prone, time-consuming and difficult to document.
Trophon2 solves this problem completely: automated, validated, reproducible — and with full documentation that provides legal certainty.

Fully automated, reproducible — under seven minutes from start to ready-for-use probe.
Place the ultrasound probe in the chamber and close the lid. The system automatically detects the probe via RFID.
approx. 30 sec.H₂O₂ ultrasound technology disinfects the entire probe surface — fully automated, with no manual steps.
approx. 5–7 min.The system automatically ventilates, and all process parameters are electronically documented and archived.
approx. 60 sec.Complete proof of disinfection in the patient record. The probe can be used directly for the next examination.
ImmediatelyWhy automated is better than manual — at a glance
Manual: variable, error-prone, not validatable
The system monitors all parameters automatically
Every cycle automatically archived — audit-proof
Staff safety through a closed system
GE · Philips · Siemens · Mindray · Samsung and others
Gynaecology · anaesthesia · cardiology · urology · radiology
The machine-based reprocessing of medical devices in Germany is governed by several guidelines and laws. Trophon2 meets all relevant requirements — and documents them completely.
As an authorised Nanosonics distributor we support you from the initial consultation through validation to training your team — personally, in Bad Kreuznach or directly at your site.
Guideline-compliant according to the recommendations of the Commission for Hospital Hygiene of the Robert Koch Institute
Guideline-compliantTrophon2 meets all requirements of the German Medical Devices Operator Ordinance (MPBetreibV) for reprocessing
MPBetreibV-compliantCE-certified and compliant with the European Medical Device Regulation
CE-certifiedRFID-based traceability of every disinfection cycle — for the hygiene department, quality assurance and audits
Audit-proofTrophon2 is compatible with a very broad range of ultrasound probes from all major manufacturers — GE Healthcare, Philips, Siemens Healthineers, Mindray, Samsung Medison and many others. We will provide a current compatibility list on request.
A complete high-level disinfection cycle takes under 7 minutes — from loading the probe to release. This makes Trophon2 significantly faster than manual disinfection methods and enables efficient probe turnover between examinations.
Trophon2 is suitable for all clinical specialities where ultrasound probes come into contact with mucous membranes: gynaecology and obstetrics, anaesthesia, cardiology, urology, radiology and interventional areas.
Validation takes place directly on site at the operator's premises — CADITEC fully supports this process. The validation documentation is generated and archived automatically.
AcuTrace PLUS uses RFID technology to automatically capture all process parameters. Every disinfection cycle is electronically documented with timestamp, probe ID, result and all relevant measured values, and can be transferred directly into hospital IT systems.
Yes. As an authorised Nanosonics distributor we support the entire rollout — from the initial demo through installation and validation to training your team. Please get in touch.
We'll show you Trophon2 in action — live at your site or with us in Bad Kreuznach.
Richard Schneider will personally get back to you.
20 minutes — by phone or video. Any questions about Trophon2, compatibility and procurement.
🔒 GDPR-compliant · German servers